Ethylene-free synthesis of polyethylene

Polyethylene (PE) is one of the most widely industrially produced and used polymers in the world. There are different types of PE, which differ in their composition of monomer units (copolymers). Depending on the molecular structure, the material behavior, such as stability and strength, varies and thus the possible applications: from films and packaging via foams, solid plastic or composite parts.
State of the art
PE is mainly produced by direct polymerization of the monomer ethylene. This requires very high pressures and temperatures to bring about polymerization for the gaseous base chemical. This technique offers only very limited possibilities for the direct polymerization of other monomers with functional groups. This limits the preparation of new copolymers and other polymer architectures.
Technology
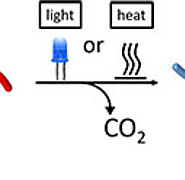
An alternative, easy to apply synthesis process has been developed by researchers from the Institute for Chemical Technology and Polymer Chemistry (ITCP). In the process, ethylene is no longer used directly as a monomer, but instead an acrylic acid ester. First, an adjustable precursor polymer, an acrylate-based solid and a targeted additional building block are synthesized in solution. The design of the precursor system already allows the subsequent structure of the polymer to be determined. The precursor is then decarboxylated in a catalytic process. This extrusion of carbon dioxide leaves a PE backbone, which is finally converted into functionalized polyethylene with the addition of a hydrogen donor. Depending on the type of additive building block, various block copolymers result, such as polystyrene-block-PE.
Advantages
With little technical effort, these synthesis steps make it possible for the first time to produce PE in a variety of copolymers and block copolymers. The polymers can be specifically functionalized with various combinations. With the aid of this method, screening experiments can be carried out quickly and inexpensively on a laboratory scale without having to handle ethylene gas.
Options for companies
The synthesis method was investigated experimentally at the institute using several examples. The method based on Decarboxylation is particularly suitable for product development in polymer chemistry. KIT is looking for industrial partners for application-specific use and further development.
Images close open
Your contact person for this offer

Innovation Manager New Materials and Health Technologies Karlsruhe Institute of Technology (KIT)
Innovation and Relations Management (IRM) Phone: +49 721 608-26107
Email: jan-niklas.bloetz@kit.edu